Introduction
Cooking is a fundamental component of food preparation associated with excessive heat, which is believed to pose health threats. This paper introduces the point of view of heat-damaged DNA in our food as a new source of genetic risks (Koszucka & Nowak, 2018). Although most studies are limited to small molecules produced during cooking and their interactions with cellular DNA, our current study will bridge this gap by examining the effect of elevated cooking temperatures on the DNA contained in the food itself.
Severe heat exposure has been associated with various health issues, including an increased risk of colorectal and pancreatic cancer, type II diabetes, cardiovascular diseases, and a decrease in life expectancy, according to Thompson and colleagues in their 2018 study. The metabolic studies of small molecules such as polycyclic aromatic hydrocarbons (PAHs) and heterocyclic amines (HCAs), which are products of particular meat, fish, and vegetable cooking, have shown that they can cumulatively produce genotoxicity and mutations after prolonged exposure (Djoumbou-Feunang et al., 2019).
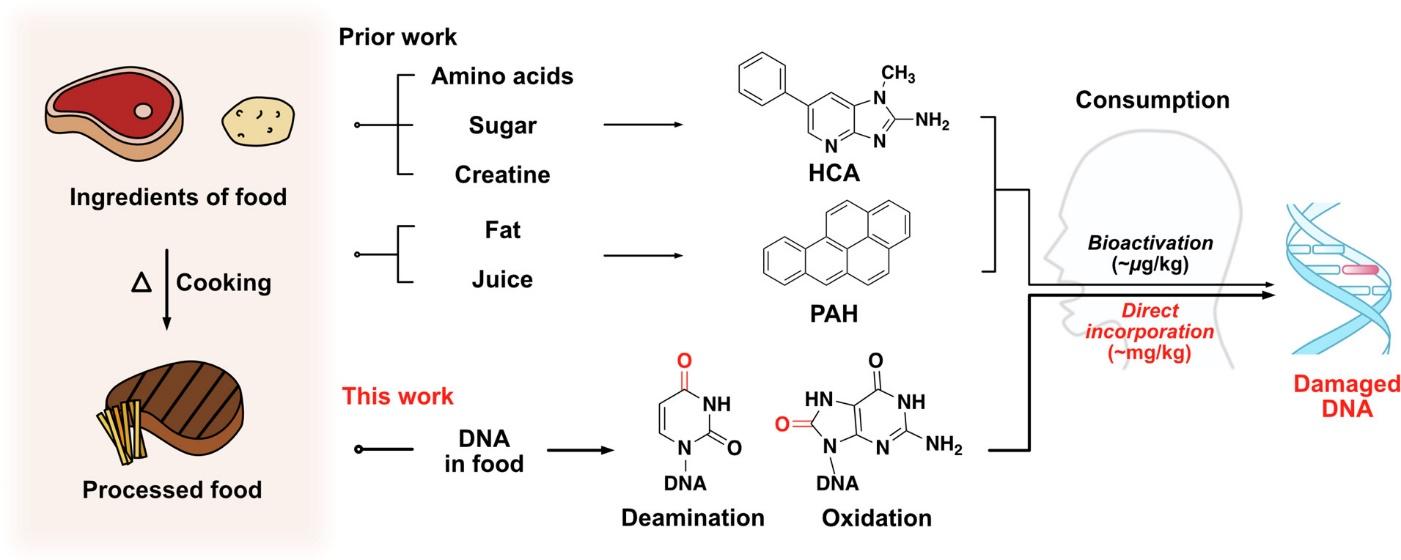
Figure 1 Heat- and air-damaged DNA in foods can be directly incorporated into mammalian tissue, potentially leading to severe DNA lesions such as mutations, abasic sites, and double-strand breaks.
The Food and Drug Administration(FDA) has already advised minimizing the consumption of red meat and deep-fried foods, which are associated with the dangers mentioned above (Sun et al., 2019). However, the present study introduced a new hypothesis –the Food DNA Damage Hypothesis. Greenberg and Bourc’his (2019) note that while DNA is a crucial macromolecule in mammalian cells, the effect of high cooking temperatures on the DNA of ingested food remains understudied.
The possible explanation for the absence of research on the DNA contained in processed food might result from the expectation that DNA from ingested food would not affect cellular processes (Deagle et al., 2018). Nevertheless, the approach will be developed further based on the assumption that the 2′-deoxyribonucleotide hydrolysis occurs when 2′-deoxynucleic acid (DNA) is fed orally. For instance, 2′-deoxynucleoside 5′-monophosphates can be dephosphorylated and taken into cells, becoming part of nucleotide salvage pathways.
The effect of high-temperature cooking on DNA stability has been validated in vitro. However, there is a need to be aware of the potential DNA damage implied when one consumes food processed by DNA-damaged food (Toyoizumi et al., 2021). Identifying how food DNA is incorporated into cellular pathways through metabolic salvage is crucial for exploring the intricate link between nutrition and gene function.
Methods
The investigation integrated control experiments, analytical approaches, and simulation models to address the research questions exhaustively. Regarding assessing the extent of DNA damage in cooked food, different food sources, including meats and vegetables with diverse DNA content, were used to represent diversity. DNA extraction was performed under controlled conditions, both in the cooked and raw food samples, to test and evaluate the degree of damage. The extent of certain types of DNA damage was determined using high-performance liquid chromatography (HPLC). This method allowed past studies to explore how cooking affects the integrity of DNA in various food products, providing insights into their interaction.
Cell lines were subjected to degraded 2′deoxynucleosides to investigate the underlying DNA damage and repair responses. Techniques like immunofluorescence, comet assays, and flow cytometry were used to analyze DNA damage and repair kinetics and chromosomal abnormalities resulting from exposure. These advanced methodologies have enabled the utilization of a broad spectrum of cellular responses that has offered insight into the underlying mechanism of the contribution of the damaged nucleosides to DNA, repair processes, and overall chromosomal stability. Exploring the consequences of damaged 2′-deoxynucleosides on the DNA damage/repair responses in the conducted experiments was performed by using a human cell line as a basic model. The study aimed to unravel the intricate mechanisms of DNA damage inducibility, its repair kinetics, and the chromosomal integrity upon nucleoside exposure by applying complicated investigative techniques like immunofluorescence, comet assays, and flow nucleoside. The applications of these advanced methodologies not only simulated the accurate responses of the cells but also demonstrated the fine-grained interactions between the damaged nucleosides and the cellular machine, which is the system responsible for maintaining genomic stability. This methodological aspect is the backbone upon which unveiling the molecular features of the DNA damage and repair processes stands.
The ultimate objective of the study was to bring together the findings derived from the multiple components of the food DNA conjecture so that a comprehensive assessment of its feasibility could be achieved. The initial observations were repeated and confirmed using diverse food varieties and different cooking methods. This process, being iterative, was directed towards consulting the scientific validity of the hypothesis. Establishing the amount of degraded DNA in preselected animal and plant tissue was the critical quantitative stage of the research. The need for animal models necessitated the adoption of strong ethical codes of conduct with attempts to guarantee subjects’ humane treatment.
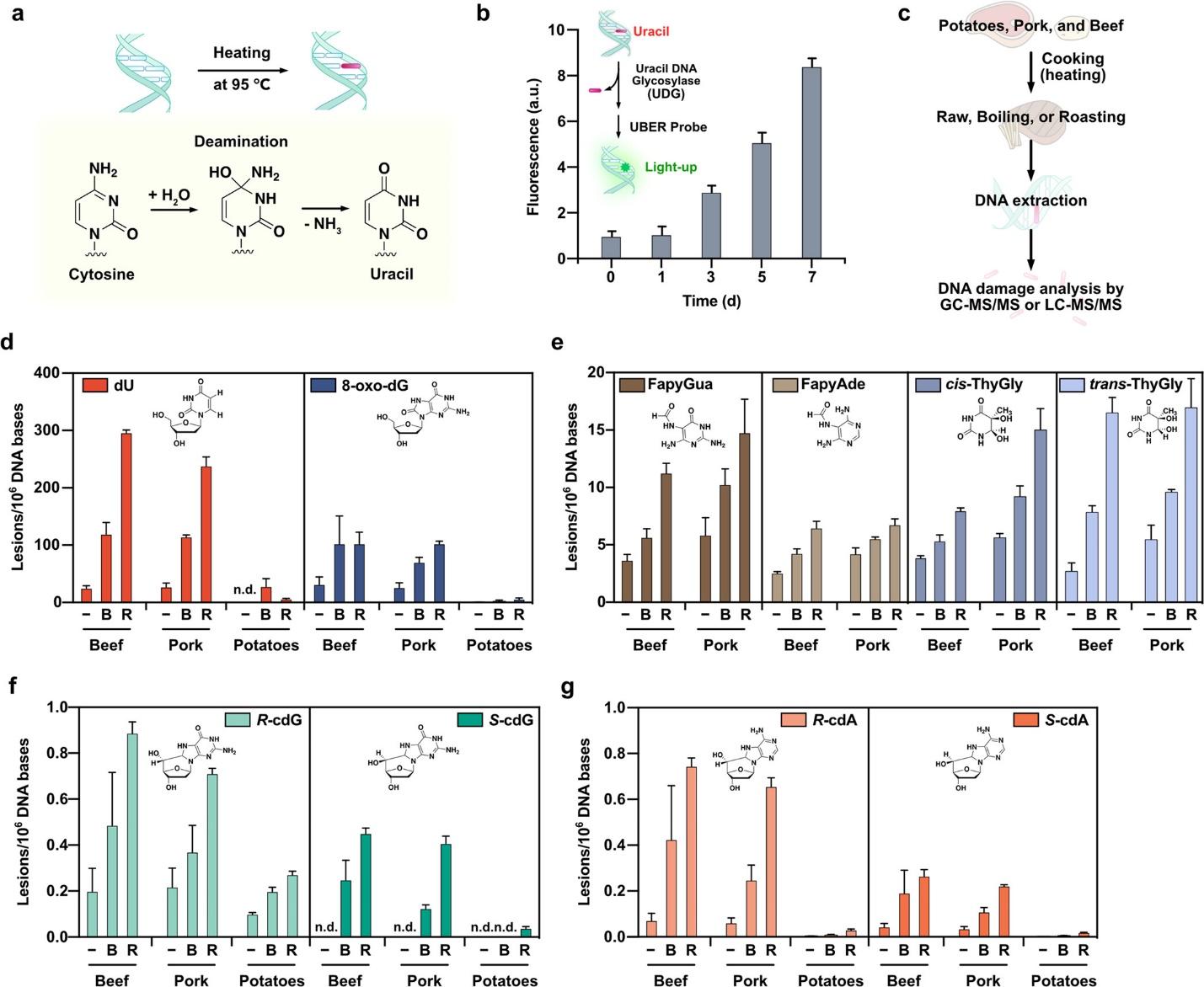
Figure 2 Elevated levels of DNA damage in food after heating and cooking. Quantification assays are used to analyze gDNA. Quantification of DNA damage levels in raw and cooked food samples
The study examines DNA damage in food from cooking methods, revealing cytosine deamination and uracil formation. DNA was extracted from potatoes, pork, and beef and subjected to raw, boiling, or roasting conditions. Quantitative assessments showed variations in DNA damage levels among beef, pork, and potatoes. The results suggest that cooking can lead to DNA alterations, potentially posing genetic risks upon consumption.
Results and Discussion
This research examined the complex relationship between culinary methods and the preservation of genomic DNA (gDNA) in food, offering significant findings regarding the repercussions of DNA damage caused by heat. Focus was made on deaminating cytosine, a widely observed type of DNA damage induced by heat; this study thoroughly investigates the effects of different culinary techniques on sliced potatoes, ground pork, and ground beef. By utilizing sophisticated scientific methods such as liquid chromatography/tandem mass spectrometry (LC-MS/MS) and gas chromatography/tandem mass spectrometry (GC-MS/MS), the research thoroughly examines the concentrations of DNA lesions in food samples that have been both uncooked and prepared. By employing this scientific methodology, a more intricate comprehension of the molecular modifications during the culinary process can be attained, thereby illuminating the hazards linked to heat-induced DNA damage in frequently consumed foods.
The study emphasizes a substantial rise in uracil concentrations in DNA under prolonged heating at high temperatures, precisely 95 °C. This increase in activity results from accelerated deamination reactions, which indicates that DNA is susceptible to damage when cooked. The disclosure is significant, given that cooking procedures frequently entail temperatures exceeding 95 °C for extended periods, which could potentially cause considerable DNA damage to the food. Moreover, this susceptibility illuminates the possible hazards linked to habitual culinary procedures. The vulnerability of DNA in food to damage increases with increasing temperature, underscoring the criticality of comprehending the intricate interplay between gastronomic inclinations and genetic soundness (Toyoizumi et al., 2021). The research’s ramifications transcend the confines of the laboratory, instigating a reassessment of culinary practices and their potential sway over human well-being.
The study reveals a significant increase in the degree of harm in foods that have undergone heat processing compared to those that have been left raw. The results concerning meats are exceptionally substantial; when exposed to roasting (at a higher temperature), ground beef and ground pork demonstrate a more pronounced increase in DNA damage than when boiled (at a lower temperature). The hallmark damage caused by DNA’s cooking process is deoxyuridine (dU) and 8-oxo-deoxyguanosine (8-oxo-dG). This highlights the specific nature of the culinary process’s impact. The observed substantial increases in these characteristic lesions following the roasting process provide insight into the varying effects of cooking methods on various DNA components. These insights are of immense value as they enhance our comprehension of the intricate molecular transformations in food due to different culinary techniques.
The investigations undertaken to assess cellular DNA damage responses yielded thought-provoking findings regarding the possible ramifications of compromised 2′deoxynucleosides on cellular mechanisms. The increase in mitochondrial base excision repair (BER) activity observed in cells incubated with damaged nucleosides indicates the presence of a cellular response mechanism striving to correct DNA lesions. The increased BER activity suggests that damaged nucleosides may have been incorporated into the structure of cellular DNA.In addition, the presence of damaged nucleosides in cells results in increased concentrations of γ-H2AX, a widely recognized biomarker for double-strand breaks (DSB), which indicates the extent of DNA damage. DSBs induce the activation of γ-H2AX, which signifies the commencement of cellular repair mechanisms. The simultaneous induction of repair mechanisms and the existence of γ-H2AX highlight the cellular endeavors to confront the difficulties of integrating compromised nucleosides. This underscores the complex relationship between DNA damage and cellular reactions.
The examination of cytotoxicity via a colony formation assay in cells exposed to damaged pyrimidines revealed noteworthy findings regarding the potential detrimental effects of heat-induced DNA damage. The colony formation assay is a dependable technique to quantify cytotoxic effects and evaluate cell viability. Damaged pyrimidines incubated with cells significantly reduced colony formation, an indicator of compromised cell viability, as determined by the study. This discovery highlights the possible adverse consequences of heat-induced DNA damage on the survival and proliferation of cells. Furthermore, chromosomal aberrations, such as chromatid gaps and exchanges, were investigated in the study as additional indicators of the detrimental consequences of nucleoside damage. The significant rise in chromosomal aberrations observed in cells exposed to damaged nucleosides underscores the possibility that heat-induced DNA damage could lead to genomic instability. Chromosomal aberrations indicate alterations in the structure of chromosomes, while their elevation signifies interference with the regular DNA replication and repair processes. By conducting a dual assessment of cytotoxicity and chromosomal aberrations, this study offers a comprehensive insight into the detrimental effects of DNA damage induced by heat, encompassing both cellular and genomic dimensions.
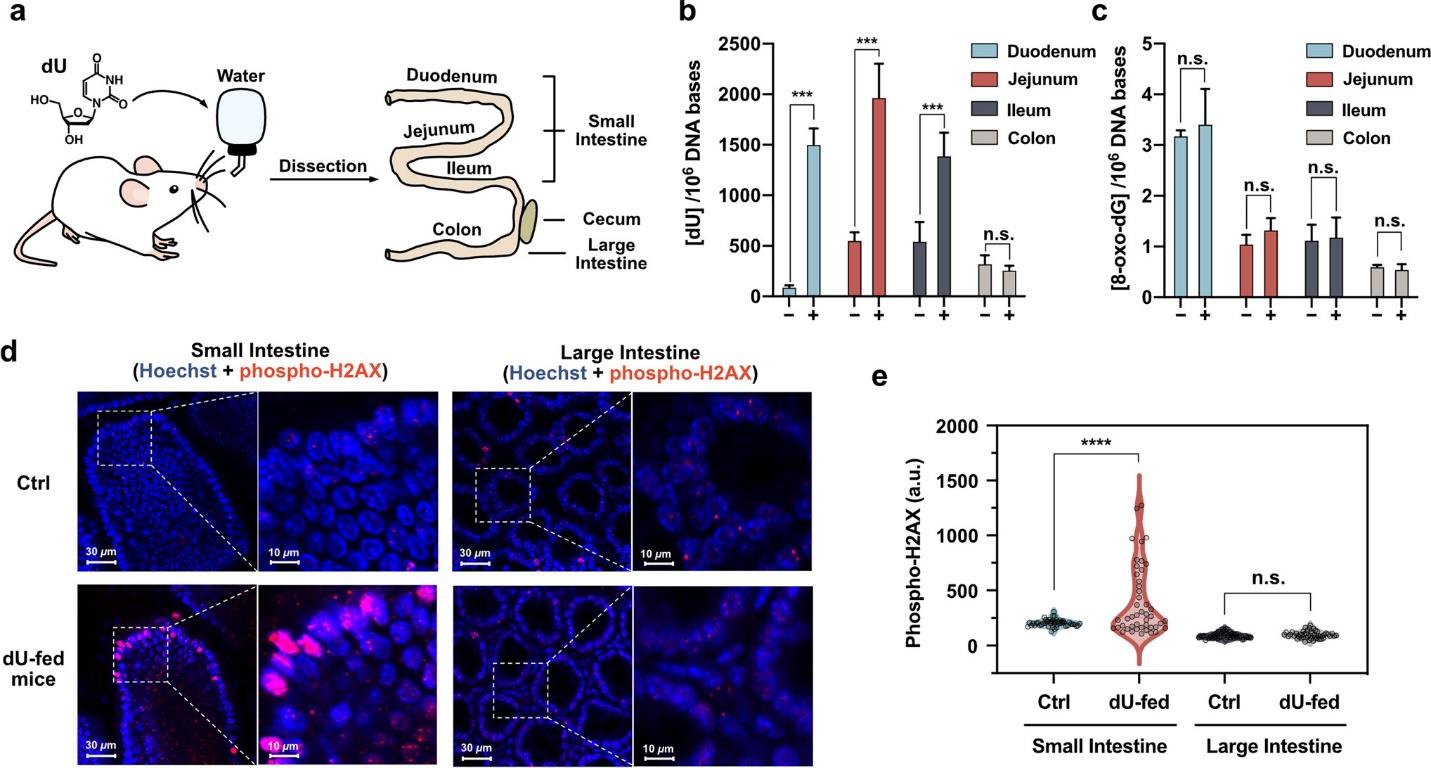
Figure 3 Genetic consequences of administering elevated concentrations of a tainted nucleoside, dU, to rodents. The subsequent impact on intestinal tissue.
The research findings demonstrate that heat-induced DNA damage leads to mutagenesis, as evidenced by significantly increased mutation levels in a hypoxanthine phosphoribosyl transferase (HPRT) mutagenesis assay triggered by damaged nucleosides, specifically dU and 8oxo-dG. As a standard method for assessing the mutagenic potential of various agents, the HPRT mutagenesis assay provides information regarding the genetic hazards associated with exposure to particular compounds. The surge in mutations that have been observed highlights the vulnerability of DNA to modifications resulting from heat-induced harm, which ultimately induces alterations in the genetic code. Mutations in critical genes, such as HPRT, can potentially contribute to the development of a range of health conditions, including cancer, by significantly impacting cellular function. Considering the broader implications for human health, this correlation between heat-induced DNA damage and mutagenesis emphasizes the significance of comprehending the potential genetic risks associated with cooking processes. Additional investigation is imperative in this field to elucidate the complete magnitude of these genetic hazards and to educate protocols for the safe preparation of foods.
The study’s findings regarding the oral gavage administration of damaged nucleosides to mice, specifically 2′-deoxyuridine (dU), provided important information regarding the possible incorporation of damaged nucleosides into cellular DNA after ingestion. The significant rise in dU concentrations observed in genomic DNA isolated from the small intestines of the mice provides compelling evidence that the impaired nucleosides were assimilated and incorporated into the cellular DNA of the gastrointestinal tissues. Furthermore, the heightened levels of γH2AX signals detected in the small intestine further confirm that dU ingestion induces increased DNA double-strand breaks. An elevated concentration of γ-H2AX, a widely recognized biomarker for DNA double-strand breaks, signifies a cellular reaction to the existence of DNA damage. The results emphasize the dynamic relationship between damaged nucleosides that have been consumed and the genomic integrity of the cells, providing insight into potential implications for the gastrointestinal system. Additional inquiries into DNA incorporation’s potential hazards and enduring consequences are imperative to attain a holistic comprehension of the matter.
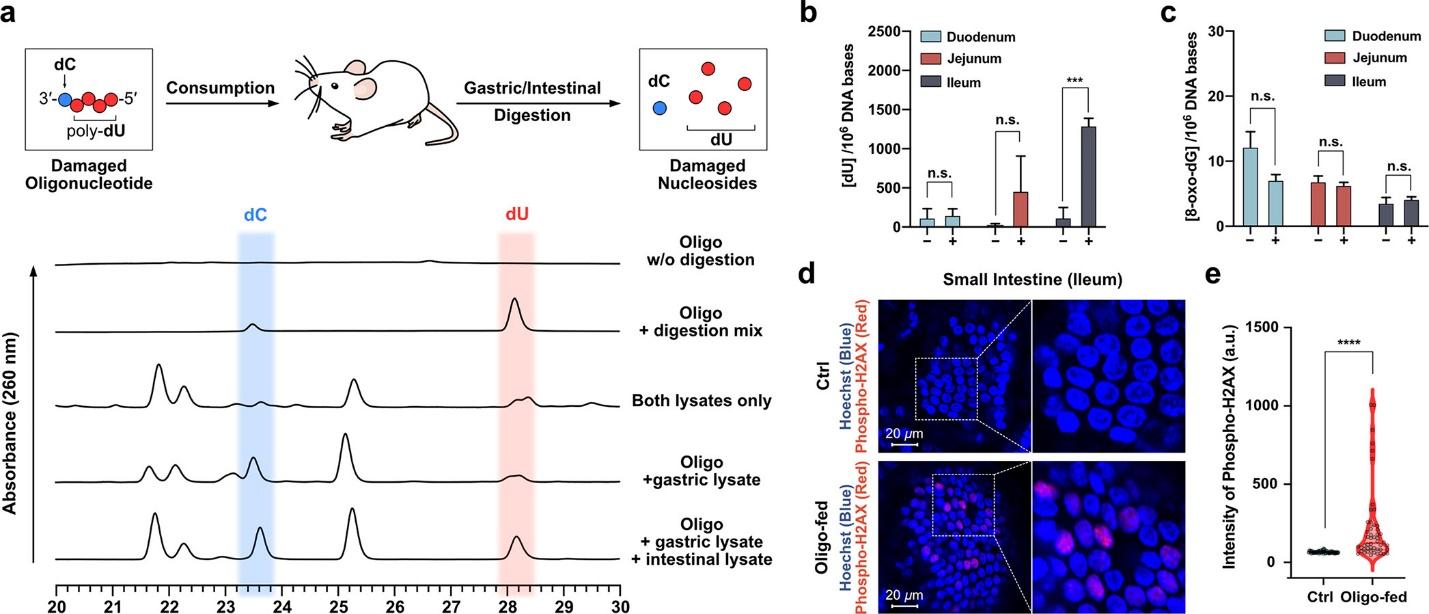
Figure 4 Examination of the metabolism of a faulty oligodeoxynucleotide in mice. It enhanced DNA double-strand break signals in small intestinal villi. It is assessed through the utilization of HPLC and immunostaining methodologies.
The study’s rigorous in vitro digestion experiments revealed that commercial digestive enzymes and gastric and intestinal lysates from mice effectively degraded damaged oligodeoxynucleotides into 2′-deoxynucleosides. The procedure above illustrated the vulnerability of compromised DNA to enzymatic digestion, thereby implying that the digestive system possesses the capacity to degrade and process DNA comprising damaged bases effectively. In the following investigation phase, rodents were administered a synthetic oligodeoxynucleotide that was compromised at its bases. A considerable increase in dU concentrations in the genomic DNA of the small intestine was a noteworthy result. The presented evidence provides substantial support for the hypothesis that damaged DNA was digested and subsequently incorporated into the cell’s genomic material. The results offer significant knowledge regarding the destiny of damaged DNA while undergoing metabolism and its possible incorporation into the DNA of the host organism. This underscores the criticality of developing a comprehensive comprehension of the ramifications for both genetic stability and health. Additional investigation in this field could shed light on the broader ramifications of ingesting compromised DNA and its interaction with the digestive mechanisms of the human organism.
This extensive investigation highlights the complex interplay between culinary procedures and the preservation of genomic DNA in food. The results emphasize the susceptibility of DNA to thermal degradation, particularly in meats cooked at elevated temperatures. The observed effects on cellular responses, cytotoxicity, chromosomal aberrations, and mutagenicity significantly heighten the genetic hazards associated with consuming heat-damaged DNA. The study underscores the necessity for additional research and considerations in food preparation, placing particular emphasis on the complex interrelationships between high-temperature cooking, dietary DNA, and potential health hazards. This study sheds light on the molecular impacts of our cooking choices in the context of evolving culinary practices.
References
Deagle, B. E., Thomas, A. C., McInnes, J. C., Clarke, L. J., Vesterinen, E. J., Clare, E. L., Kartzinel, T. R., & Eveson, J. P. (2018). Counting with <scp>DNA</scp> in metabarcoding studies: How should we convert sequence reads to dietary data? Molecular Ecology, 28(2), 391-406. https://doi.org/10.1111/mec.14734
Djoumbou-Feunang, Y., Fiamoncini, J., Gil-de-la-Fuente, A., Greiner, R., Manach, C., & Wishart, D. S. (2019). BioTransformer: A comprehensive computational tool for small molecule metabolism prediction and metabolite identification. Journal of Cheminformatics, 11(1). https://doi.org/10.1186/s13321-018-0324-5
Greenberg, M. V., & Bourc’his, D. (2019). The diverse roles of DNA methylation in mammalian development and disease. Nature Reviews Molecular Cell Biology, 20(10), 590–607. https://doi.org/10.1038/s41580-019-0159-6
Koszucka, A., & Nowak, A. (2018). Thermal processing food-related toxicants: A review. Critical Reviews in Food Science and Nutrition, 59(22), 3579-3596. https://doi.org/10.1080/10408398.2018.1500440
Sun, Y., Liu, B., Snetselaar, L. G., Robinson, J. G., Wallace, R. B., Peterson, L. L., & Bao, W. (2019). Association of fried food consumption with all-cause, cardiovascular, and cancer mortality: Prospective cohort study. BMJ, k5420. https://doi.org/10.1136/bmj.k5420
Thompson, R., Hornigold, R., Page, L., & Waite, T. (2018). Associations between high ambient temperatures and heat waves with mental health outcomes: A systematic review. Public Health, 161, 171-191. https://doi.org/10.1016/j.puhe.2018.06.008
Toyoizumi, T., Kosugi, T., Toyama, Y., & Nakajima, T. (2021). Effects of high-temperature cooking on the gamma-aminobutyric acid content and antioxidant capacity of germinated brown rice (Oryza et al.). CyTA – Journal of Food, 19(1), 360-369. https://doi.org/10.1080/19476337.2021.1905721
 write
write